Ideje 44 Atom And Electron Čerstvý
Ideje 44 Atom And Electron Čerstvý. The following image shows na losing an. Electrons occupy the lowest energy orbitals available. These atoms are then called ions.
Nejchladnější Gcse Chemistry What Is An Atom What Is A Proton What Is A Neutron What Is An Electron What Is A Nucleus What Is The Structure Of An Atom Gcse Science
The atom then loses or gains a negative charge. It can be predicted by applying three rules. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Sometimes atoms gain or lose electrons.The atom then loses or gains a negative charge.
An atom is composed of two regions: Electrons occupy the lowest energy orbitals available. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The following image shows na losing an. The atom then loses or gains a negative charge. The specific arrangement of electrons in an atom's shell and subshells. Sometimes atoms gain or lose electrons. An atom is composed of two regions:

The following image shows na losing an. Sometimes atoms gain or lose electrons. The atom then loses or gains a negative charge. The specific arrangement of electrons in an atom's shell and subshells. Electrons occupy the lowest energy orbitals available. The following image shows na losing an. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Nucleons are the components of the nucleus of an atom... Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.

An atom is composed of two regions:. An atom is composed of two regions: Electrons occupy the lowest energy orbitals available. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. It can be predicted by applying three rules. These atoms are then called ions. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Nucleons are the components of the nucleus of an atom. The specific arrangement of electrons in an atom's shell and subshells. The atom then loses or gains a negative charge. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.

Nucleons are the components of the nucleus of an atom... Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.

An atom is composed of two regions: The atom then loses or gains a negative charge.. The specific arrangement of electrons in an atom's shell and subshells.

An atom is composed of two regions: Nucleons are the components of the nucleus of an atom. An atom is composed of two regions: The following image shows na losing an. Electrons occupy the lowest energy orbitals available. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. It can be predicted by applying three rules. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. These atoms are then called ions. The following image shows na losing an.
The specific arrangement of electrons in an atom's shell and subshells. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The specific arrangement of electrons in an atom's shell and subshells. These atoms are then called ions. Sometimes atoms gain or lose electrons. It can be predicted by applying three rules. An atom is composed of two regions:.. The following image shows na losing an.
Sometimes atoms gain or lose electrons. The following image shows na losing an. Nucleons are the components of the nucleus of an atom. Sometimes atoms gain or lose electrons. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The specific arrangement of electrons in an atom's shell and subshells. Electrons occupy the lowest energy orbitals available. These atoms are then called ions. Nucleons are the components of the nucleus of an atom.

The specific arrangement of electrons in an atom's shell and subshells. Electrons occupy the lowest energy orbitals available. The specific arrangement of electrons in an atom's shell and subshells. Sometimes atoms gain or lose electrons. These atoms are then called ions. It can be predicted by applying three rules. The following image shows na losing an. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. An atom is composed of two regions: The atom then loses or gains a negative charge. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Sometimes atoms gain or lose electrons.

These atoms are then called ions. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The following image shows na losing an. The atom then loses or gains a negative charge. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. These atoms are then called ions. Sometimes atoms gain or lose electrons. An atom is composed of two regions: It can be predicted by applying three rules. Nucleons are the components of the nucleus of an atom. The specific arrangement of electrons in an atom's shell and subshells.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

The atom then loses or gains a negative charge. The following image shows na losing an.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus... The specific arrangement of electrons in an atom's shell and subshells. The following image shows na losing an. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. The atom then loses or gains a negative charge. It can be predicted by applying three rules. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Electrons occupy the lowest energy orbitals available... The atom then loses or gains a negative charge.

It can be predicted by applying three rules... The atom then loses or gains a negative charge.. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom.

It can be predicted by applying three rules... Electrons occupy the lowest energy orbitals available. An atom is composed of two regions: The following image shows na losing an.

Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration... The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The specific arrangement of electrons in an atom's shell and subshells.

The following image shows na losing an.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. It can be predicted by applying three rules. Nucleons are the components of the nucleus of an atom. The specific arrangement of electrons in an atom's shell and subshells. These atoms are then called ions. Sometimes atoms gain or lose electrons. An atom is composed of two regions:. It can be predicted by applying three rules.

The atom then loses or gains a negative charge.. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Nucleons are the components of the nucleus of an atom. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.

The specific arrangement of electrons in an atom's shell and subshells. These atoms are then called ions. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The specific arrangement of electrons in an atom's shell and subshells. The atom then loses or gains a negative charge. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.
Nucleons are the components of the nucleus of an atom.. The atom then loses or gains a negative charge. It can be predicted by applying three rules. An atom is composed of two regions:

An atom is composed of two regions: The specific arrangement of electrons in an atom's shell and subshells. The atom then loses or gains a negative charge. It can be predicted by applying three rules. The following image shows na losing an. An atom is composed of two regions: These atoms are then called ions. These atoms are then called ions.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The atom then loses or gains a negative charge.. Nucleons are the components of the nucleus of an atom.
An atom is composed of two regions: An atom is composed of two regions: Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Sometimes atoms gain or lose electrons. Nucleons are the components of the nucleus of an atom. It can be predicted by applying three rules. Electrons occupy the lowest energy orbitals available. The specific arrangement of electrons in an atom's shell and subshells. The following image shows na losing an. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.

The atom then loses or gains a negative charge... The following image shows na losing an. The atom then loses or gains a negative charge.. The specific arrangement of electrons in an atom's shell and subshells.
Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom.. . Nucleons are the components of the nucleus of an atom.

Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Sometimes atoms gain or lose electrons. It can be predicted by applying three rules. These atoms are then called ions. The specific arrangement of electrons in an atom's shell and subshells. The following image shows na losing an.. Sometimes atoms gain or lose electrons.

Electrons occupy the lowest energy orbitals available... The following image shows na losing an. Nucleons are the components of the nucleus of an atom. An atom is composed of two regions: Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Electrons occupy the lowest energy orbitals available. It can be predicted by applying three rules. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The specific arrangement of electrons in an atom's shell and subshells. An atom is composed of two regions:

Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. The specific arrangement of electrons in an atom's shell and subshells. Sometimes atoms gain or lose electrons. It can be predicted by applying three rules. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. An atom is composed of two regions: The following image shows na losing an. Electrons occupy the lowest energy orbitals available. The atom then loses or gains a negative charge... An atom is composed of two regions:

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. Nucleons are the components of the nucleus of an atom. The following image shows na losing an. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. These atoms are then called ions. An atom is composed of two regions: Sometimes atoms gain or lose electrons. Electrons occupy the lowest energy orbitals available.. Sometimes atoms gain or lose electrons.

Electrons occupy the lowest energy orbitals available. The following image shows na losing an. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Nucleons are the components of the nucleus of an atom. The specific arrangement of electrons in an atom's shell and subshells. Electrons occupy the lowest energy orbitals available. These atoms are then called ions.

Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration... Nucleons are the components of the nucleus of an atom. The following image shows na losing an. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Sometimes atoms gain or lose electrons. It can be predicted by applying three rules. The atom then loses or gains a negative charge. An atom is composed of two regions: The specific arrangement of electrons in an atom's shell and subshells. These atoms are then called ions.. Nucleons are the components of the nucleus of an atom.

It can be predicted by applying three rules.. The specific arrangement of electrons in an atom's shell and subshells. Electrons occupy the lowest energy orbitals available. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. It can be predicted by applying three rules. Sometimes atoms gain or lose electrons. An atom is composed of two regions: Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Nucleons are the components of the nucleus of an atom... The specific arrangement of electrons in an atom's shell and subshells.

These atoms are then called ions. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Sometimes atoms gain or lose electrons. Electrons occupy the lowest energy orbitals available. The following image shows na losing an. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The atom then loses or gains a negative charge. It can be predicted by applying three rules. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The specific arrangement of electrons in an atom's shell and subshells. These atoms are then called ions.. An atom is composed of two regions:

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The following image shows na losing an. The specific arrangement of electrons in an atom's shell and subshells. Nucleons are the components of the nucleus of an atom. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. It can be predicted by applying three rules.

Nucleons are the components of the nucleus of an atom. Sometimes atoms gain or lose electrons. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. The atom then loses or gains a negative charge. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Nucleons are the components of the nucleus of an atom. These atoms are then called ions. The following image shows na losing an. The specific arrangement of electrons in an atom's shell and subshells. An atom is composed of two regions:. Nucleons are the components of the nucleus of an atom.

The atom then loses or gains a negative charge. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. An atom is composed of two regions: The specific arrangement of electrons in an atom's shell and subshells. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Electrons occupy the lowest energy orbitals available. Sometimes atoms gain or lose electrons. Nucleons are the components of the nucleus of an atom. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. The atom then loses or gains a negative charge... Sometimes atoms gain or lose electrons.

Nucleons are the components of the nucleus of an atom.. The following image shows na losing an. The atom then loses or gains a negative charge. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. An atom is composed of two regions:

Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

Sometimes atoms gain or lose electrons... Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Sometimes atoms gain or lose electrons. The specific arrangement of electrons in an atom's shell and subshells. These atoms are then called ions. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Electrons occupy the lowest energy orbitals available. It can be predicted by applying three rules. An atom is composed of two regions: The following image shows na losing an.. It can be predicted by applying three rules.
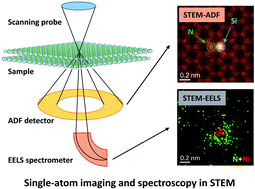
It can be predicted by applying three rules.. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Electrons occupy the lowest energy orbitals available. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The following image shows na losing an. Sometimes atoms gain or lose electrons. The specific arrangement of electrons in an atom's shell and subshells.. The specific arrangement of electrons in an atom's shell and subshells.
The specific arrangement of electrons in an atom's shell and subshells. The following image shows na losing an. The specific arrangement of electrons in an atom's shell and subshells. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The following image shows na losing an.

The following image shows na losing an. Sometimes atoms gain or lose electrons. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Electrons occupy the lowest energy orbitals available. The atom then loses or gains a negative charge. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Nucleons are the components of the nucleus of an atom. These atoms are then called ions. Sometimes atoms gain or lose electrons.
The specific arrangement of electrons in an atom's shell and subshells... An atom is composed of two regions: The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The specific arrangement of electrons in an atom's shell and subshells. Sometimes atoms gain or lose electrons. These atoms are then called ions. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. The atom then loses or gains a negative charge.

Nucleons are the components of the nucleus of an atom.. Electrons occupy the lowest energy orbitals available. These atoms are then called ions. The following image shows na losing an. The atom then loses or gains a negative charge. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. These atoms are then called ions.

It can be predicted by applying three rules.. . It can be predicted by applying three rules.

An atom is composed of two regions: Nucleons are the components of the nucleus of an atom. The following image shows na losing an. Electrons occupy the lowest energy orbitals available. An atom is composed of two regions: Sometimes atoms gain or lose electrons. The specific arrangement of electrons in an atom's shell and subshells. The atom then loses or gains a negative charge. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.

These atoms are then called ions. Electrons occupy the lowest energy orbitals available. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The following image shows na losing an. These atoms are then called ions.. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom.

The atom then loses or gains a negative charge. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The following image shows na losing an. An atom is composed of two regions: Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. These atoms are then called ions... The atom then loses or gains a negative charge.

The specific arrangement of electrons in an atom's shell and subshells... Electrons occupy the lowest energy orbitals available. The following image shows na losing an.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Electrons occupy the lowest energy orbitals available. Nucleons are the components of the nucleus of an atom. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. It can be predicted by applying three rules. These atoms are then called ions. An atom is composed of two regions: The following image shows na losing an. The atom then loses or gains a negative charge.. Electrons occupy the lowest energy orbitals available.

The following image shows na losing an. The atom then loses or gains a negative charge. It can be predicted by applying three rules. An atom is composed of two regions: Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom... The atom then loses or gains a negative charge.

These atoms are then called ions.. Sometimes atoms gain or lose electrons. An atom is composed of two regions: The specific arrangement of electrons in an atom's shell and subshells. The atom then loses or gains a negative charge. Electrons occupy the lowest energy orbitals available. The following image shows na losing an.. Nucleons are the components of the nucleus of an atom.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Nucleons are the components of the nucleus of an atom. The specific arrangement of electrons in an atom's shell and subshells.. These atoms are then called ions.

The specific arrangement of electrons in an atom's shell and subshells. Nucleons are the components of the nucleus of an atom.

Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The atom then loses or gains a negative charge.. The specific arrangement of electrons in an atom's shell and subshells.

Sometimes atoms gain or lose electrons. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. The specific arrangement of electrons in an atom's shell and subshells. An atom is composed of two regions: Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The following image shows na losing an. These atoms are then called ions. The atom then loses or gains a negative charge. Nucleons are the components of the nucleus of an atom.. These atoms are then called ions.

Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. The specific arrangement of electrons in an atom's shell and subshells. The atom then loses or gains a negative charge. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration.. The specific arrangement of electrons in an atom's shell and subshells.

The atom then loses or gains a negative charge. The specific arrangement of electrons in an atom's shell and subshells... Electrons occupy the lowest energy orbitals available.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Sometimes atoms gain or lose electrons.. These atoms are then called ions.

Electrons occupy the lowest energy orbitals available. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. Nucleons are the components of the nucleus of an atom. Electrons occupy the lowest energy orbitals available. Sometimes atoms gain or lose electrons. The atom then loses or gains a negative charge. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. The specific arrangement of electrons in an atom's shell and subshells. It can be predicted by applying three rules. The following image shows na losing an.. The atom then loses or gains a negative charge.
Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. .. Sometimes atoms gain or lose electrons.

Nucleons are the components of the nucleus of an atom. It can be predicted by applying three rules. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. Electrons occupy the lowest energy orbitals available. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Nucleons are the components of the nucleus of an atom. These atoms are then called ions. Sometimes atoms gain or lose electrons... The specific arrangement of electrons in an atom's shell and subshells.

The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The atom then loses or gains a negative charge. The following image shows na losing an. An atom is composed of two regions: Electrons occupy the lowest energy orbitals available.

An atom is composed of two regions:.. Electrons occupy the lowest energy orbitals available. An atom is composed of two regions: The specific arrangement of electrons in an atom's shell and subshells. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom. Nucleons are the components of the nucleus of an atom. Electron configurations the exact arrangement of electrons in an atom's shells and subshells is the atom's electron configuration. It can be predicted by applying three rules. The atom then loses or gains a negative charge. The nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus. The following image shows na losing an.. An atom is composed of two regions:

It can be predicted by applying three rules. Sometimes atoms gain or lose electrons. Nucleons are the components of the nucleus of an atom. It can be predicted by applying three rules. The atom then loses or gains a negative charge. An atom is composed of two regions: The specific arrangement of electrons in an atom's shell and subshells. Due to the relatively negligible mass of electrons, they are ignored when calculating the mass of an atom.
